By Erin Hayes-Pontius
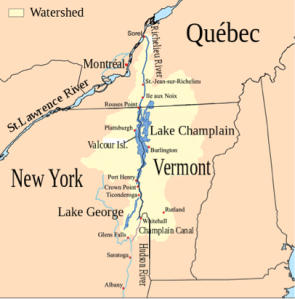
Paleoecology, or the study of organisms and their environments in past times, has a specific branch known as paleolimnology, which deals with records left by lakes. Beyond being pleasant places for swimming, fishing, and other recreation, lakes are particularly valuable for paleo records. Not only do lakes archive the goings-on within themselves, but they also archive goings-on within their drainage basins. In the case of Lake Champlain, it has a drainage basin nearly 17 times that of its surface area (21,326 km2 : 1,269 km2), making it extra sensitive to changes in climate (Fig. 1).
Due to lakes’ positions at low points on the landscape, they integrate everything that happens within their drainage basin and are therefore important “sentinels of change” (Williamson et al. 2009).
Over time, a variety of things fall into lakes, such as diatoms (single-celled algae), pollen, chironomid midges, or needles, to name a few, and these things get buried by sediment. In some lakes- some, certainly not most- sediment deposits occur quickly enough to leave annual layers; if this interests you, definitely check out Rob’s post from a little while back. Unfortunately, this does not happen most of the time, and we’re left with a much coarser resolution to deal with. Rather than knowing about the diatom species every year, we may only know ‘who’ was there every few years, every decade, or even every few decades. Despite these challenges, lakes are still- and will continue to be- widely used in paleoecology.
In order to interpret environmental changes from the proxy of your choosing, there are a few ways to go about it. Early on, some paleolimnologists categorized various species into different ‘groups’ or along a continuum of some variable based on their distributions. One such early- and famous- example of this was Hustedt’s categorization of diatoms into different pH groups (1939). From that starting point, were you to graph species composition over time, you could see which diatoms increased, decreased, or did not respond to various time periods and make inferences about the value of some environmental variable, such as pH. For example, if you noticed acid-loving species becoming more prevalent in the past, you might think to yourself, “Oh, that must be from the increase in acid rain during that time period. Now we know that pristine lakes were probably affected by human actions hundreds of miles away!” Of course, you could apply this process to other variables of interest, such as salinity, phosphorus, nitrogen, or temperature, to name a few.
Not content to study lakes on such qualitative terms, paleolimnologists took these inferences a step further and made an attempt at making environment-diatom relationships quantitative. This new quantitative method used what is called a transfer function to establish a numerical value of the variable in question, such as pH, based on the pH optima of present diatoms in a given lake (Oksanen et al. 1988). Transfer functions are typically based off of a large set of lakes exhibiting a wide range of the variable in question; for a salinity transfer function, you would want to include fresh lakes, salty lakes, and of course, many along the continuum in between the two extremes. Below is an extremely simplified version of how transfer functions are made (Fig. 2).
Figure 3. Fundamental, potential, and realized niches. Crudely drawn in Powerpoint by the author; see Jackson and Overpeck 2000 for the original version of this figure.
Unfortunately, transfer functions are not fool-proof, and there are some serious pitfalls to using them (see Juggins 2013 for an exhaustive review). The saying “A little knowledge is a dangerous thing” is no exception here. First off, when people develop transfer functions, they conducted a survey to obtain their data- they (most likely) did not conduct experiments. Maybe this doesn’t sound too terrible to you, but think of it this way: did the diatom species occur most often at that particular concentration of phosphorus because of the phosphorus concentration, or is it some other factor altering its abundance? Until you run an experiment and manipulate the phosphorus concentration to see what the ideal concentration is, you will not and cannot presume to know what that species’ preference is since some other variable might be at work.
A second problem is that the species- environment relationship, or the niche, may not hold up through time. While there is some evidence that niches may be conserved, it is still a caveat that we have to take into consideration. For example, it is entirely possible that a species can exist in some combination of environmental variables that isn’t even present today! Let me back up, since that may have been too big of a leap. When I grew up in ecology, we learned about two types of species niches (the specific set of environmental conditions a species requires): the fundamental niche, which is the entire range of present habitat a species can live in, and the realized niche, which is where the species lives when you take competition from other species into account. Another type of niche that I only heard about recently, the potential niche, refers to sets of conditions that don’t exist right now, but could support the species (Fig. 3).
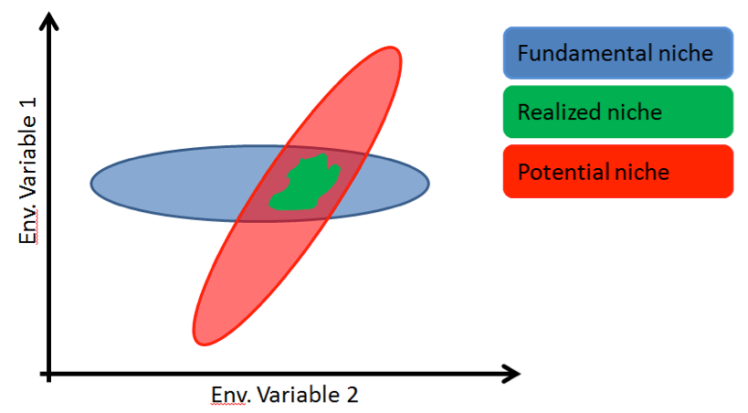
Figure 3. Fundamental, potential, and realized niches. Crudely drawn in Powerpoint by the author; see Jackson and Overpeck 2000 for the original version of this figure.
Taking this concept back to paleolimnology, what if you see a particular indicator species and make an erroneous assumption about the environment? You might infer that the environmental variable of interest must have been somewhere in the green patch at that time period, when in fact it could have been anywhere inside that red patch (Fig. 3). To further complicate matters, what if competitive interactions between species changed over time, thus altering the realized niche space between then and now?
What’s a paleoecologist to do?
Well, calling it quits is an option, but no one really wants that; we end up doing the best we can with what we know. To avoid making erroneous assumptions about the ecology of study species, we need to combine paleolimnological studies with modern limnological experiments (Saros et al. 2012). To do this, we need to actually manipulate variables of interest and see how the species responds- either by growth rate, biovolume, density, etc. Better yet: see how different variables, such as light and nutrients, interact by carefully designing the experiments. Then, we can make some more useful, and hopefully accurate, inferences about past climate.
Works Cited
Hustedt F., 1939. Systematische und okologische Untersuchungentiber die Diatomeenflora von Java, Bali undSumatra. Arch. Hydrobiol./Suppl. 16: 274-394.
Jackson ST, Overpeck JT. 2000. Responses of plant populations and communities to environmental changes of the Late Quaternary. Paleobiology 26(4):194-220.
Oksanen J, Läärä E, Huttenun P, Meriläinen J. 1988. Estimation of pH optima and tolerances of diatoms in lake sediments by the methods of weighted averaging, least squares and maximum likelihood, and their use for the prediction of lake acidity. Journal of Paleolimnology 1(1):39-49.
Saros JE, Stone JR, Pederson GT, Slemmons KEH, Spanbauer T, Schliep A, Cahl D, Williamson CE, Engstrom DR. 2012. Climate-induced changes in lake ecosystem structure inferred from coupled neo- and paleoecological approaches. Ecology 93(10):2155-2164.
Williamson CE, Saros JE, Schindler DW. 2009. Sentinels of Change. Science 323:887-888.

I’d argue that perhaps calling it quits is the right course of action, at least as far as current transfer functions go. The big elephant in the room is that even if you had a perfect model of environmental requirements of all your species built from a perfect data set with no environmental noise in it such that we perfectly capture the species-environment relationship of interest, you still have to apply it downcore where you have no control over what went on. Even with the sorts of neo- palaeolimnological combinations of the sorts Jasmine et al describe, our current approaches offer little more than semi-quantitative or qualitative reconstructions; we might use our neolimnological information to “constrain” or validate the reconstruction but the reconstruction itself is still very much based on our crude technology from the 1980s and 90s.
If the variable of interest is ecologically important and the sequence/time period you study contains a sufficiently large response to the variable of interest then the transfer function reconstruction more than likely will give you a reconstruction where the trend or pattern of change is about right, perhaps with reasonable representation of the magnitude of the change. If you are very lucky the actual reconstructed values might be reliable and accurate. Many of the applications of transfer functions however are now focussing on a range of environmental perturbations, many of which don’t change by large amounts. Are those reconstructions reliable or even useful? I don’t know. I doubt all are…
I’m not suggesting we throw the quantitative baby out with the bathwater though. A transfer function is very much like an SDM (Species Distribution Model); we stack information from individual species’ responses to the environment, responses that reflect the realized niche etc. There has been a lot of gnashing of teeth and wailing over SDMs of late but we are starting to see more advanced models for joint species distributions. Inverting one of those models (as we do in the Maximum Likelihood / Gaussian Logit Regression transfer function methods) to give some estimate of the past environment from composition would be a most useful exercise and likely lead to an considerable advance in the field.
Thank you for your comment- I’m wicked new to paleoecology, so this post is me trying to make sense of transfer functions. Do you have any readings on SDMs or their inversions? I’d love to read up on other paleo methods!